Fluorescence sensing of Cu2+ ion and imaging of fungal cell by ultra-small fluorescent carbon dots derived from Acacia concinna seeds - ScienceDirect

SOLVED: Part B Cu2+ or Cd2+ Ion Identificatlon For the reactions shown below; we added 3.75 mL of 0.0410 M NazS to test tube containing one of the two cations (Cuzt or

Reactivity-Based Detection of Copper(II) Ion in Water: Oxidative Cyclization of Azoaromatics as Fluorescence Turn-On Signaling Mechanism | Journal of the American Chemical Society

a The tetra coordinated Cu²⁺ ion in 2; b the binuclear {Cu2} secondary... | Download Scientific Diagram

Stabilizing Cu2+ Ions by Solid Solutions to Promote CO2 Electroreduction to Methane | Journal of the American Chemical Society

a) Coordination environments of Cu1 and Cu2 ions in 1. Symmetry codes:... | Download Scientific Diagram

Why is Cu+ diamagnetic while Cu2+ is paramagnetic? | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium

Determination of new Cu+, Cu2+, and Zn2+ Lennard-Jones ion parameters in acetonitrile. | Semantic Scholar
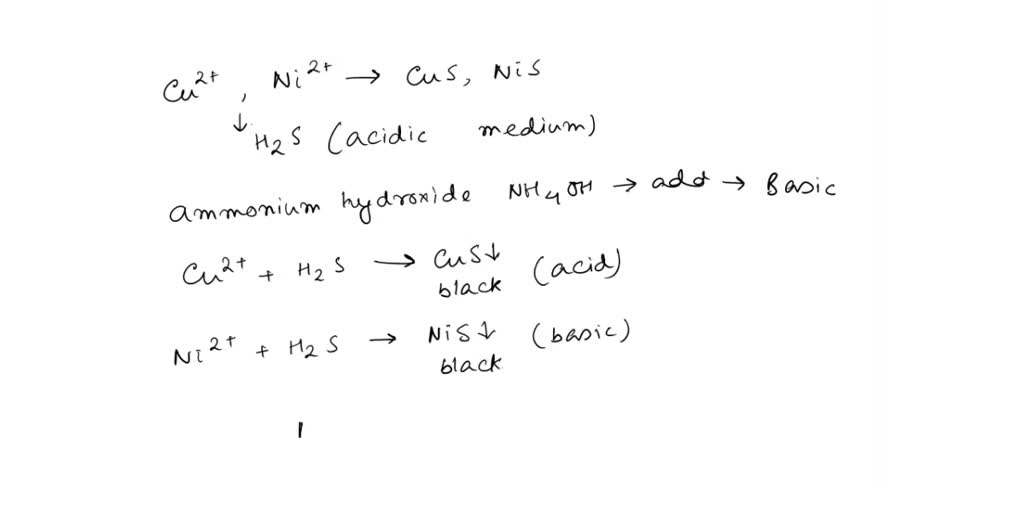
SOLVED: If a solution contains Cu2+ and Ni2+ ions, what test reagent can be used to separate these ions? Write equations indicating the results.

Copper Cu transition metal Chemistry copper(I) Cu+ copper(II) Cu2+ ion complex ions ligand substitution compounds redox chemical reactions principal oxidation states +1 +2 GCE AS A2 IB A level inorganic chemistry revision
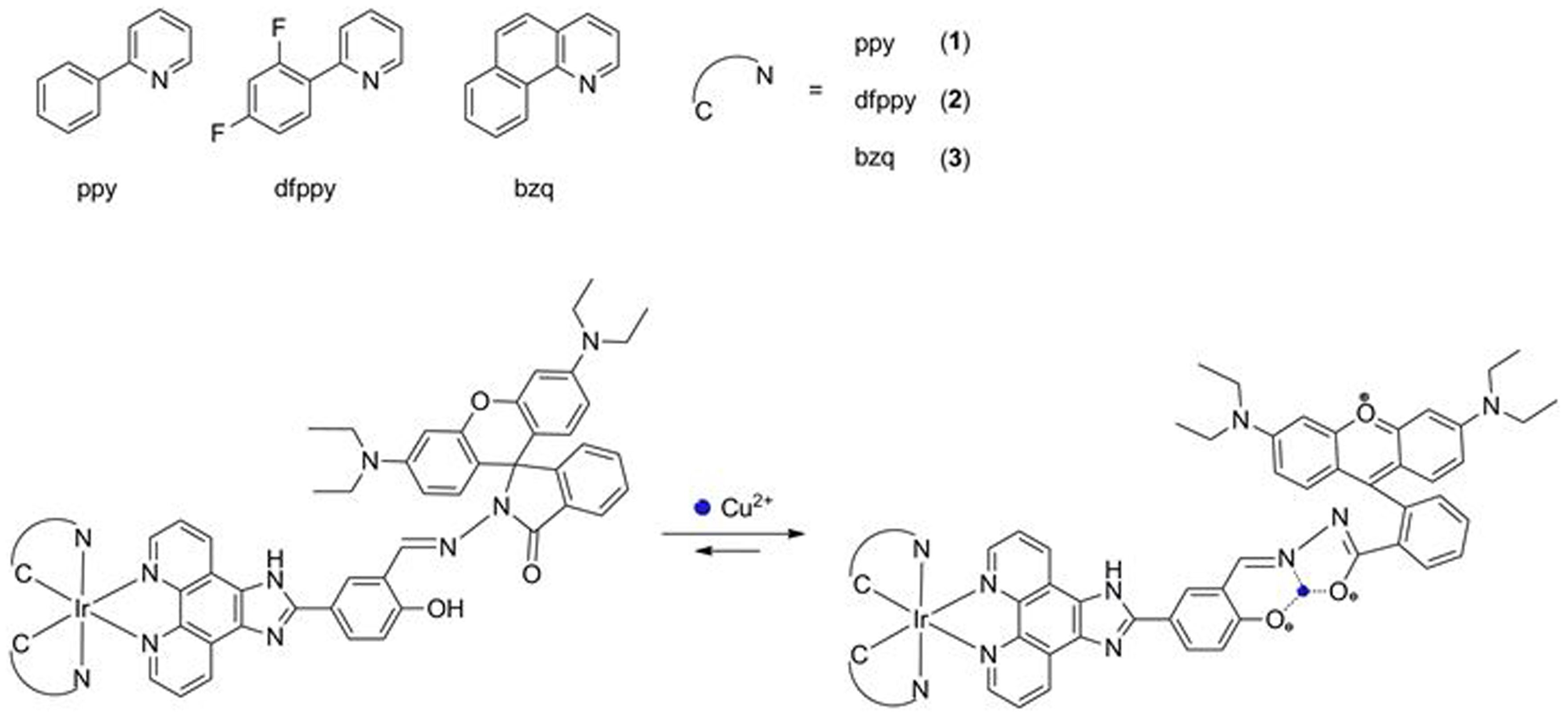




.jpg)